|
 Scientists
Discover New Gene Essential for the Development Of Normal Brain
Connections Resulting From Sensory Input Scientists
Discover New Gene Essential for the Development Of Normal Brain
Connections Resulting From Sensory Input
by Sherry Seethaler, University of Southern California,
January 8, 2004
For more articles like this
visit
https://www.bridges4kids.org.
Biologists at
the University of California, San Diego and the Johns Hopkins
University have discovered a gene that plays a key role in
initiating changes in the brain in response to sensory
experience, a finding that may provide insight into certain
types of learning disorders.
After birth, learning and experience change the architecture of
the brain dramatically. The structure of individual neurons, or
nerve cells, changes during learning to accommodate new
connections between neurons. Neuroscientists believe these
structural changes are initiated when neurons are activated,
causing calcium ions to flow into cells and alter the activity
of genes.
In a paper featured on the cover of the January 9th issue of the
journal Science, biologists at UCSD and the Johns Hopkins
University medical school report the discovery of the first
gene, CREST, known to mediate these changes in the structure of
neurons in response to calcium.
“We discovered the gene CREST using a new method we developed to
identify genes that are switched on in the presence of calcium,”
says Anirvan Ghosh, a professor of biology at UCSD who headed
the study. “The brains of mice lacking CREST appear normal at
birth, but do not develop normally in response to sensory
experience after birth. This parallels some learning disorders
in humans where the child appears normal initially, but by the
age of two or three years it becomes clear that there are
failures in the acquisition of new knowledge.”
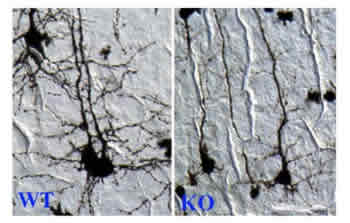 |
|
Images of
neurons from normal mice (left) and from mice lacking CREST gene
(right)
Credit:
Anirvan Ghosh |
Neurons from normal mice develop a highly branched tree-like
structure. In fact, much of the growth of the brain that occurs
soon after birth is the development and branching of
dendrites—the part of a nerve cell that receives input from
other neurons. Thus, this branching allows neurons to form many
different synapses, or connections, with many other neurons,
permitting much cross talk between them. Neurons taken from mice
lacking the CREST gene are more linear, like a plant shoot.
In addition, when individual neurons kept alive in a Petri dish
are stimulated with calcium ions, they respond by developing
highly branched dendrites, but neurons taken from mice lacking
CREST fail to branch in response to calcium.
“CREST is the first example of a transcription factor—a protein
that turns genes on and off—that appears to be specifically
required for the development of brain neurons after birth,"
explains Ghosh, who conducted the study at his former laboratory
at Johns Hopkins.
His new laboratory at UCSD is currently working to determine
what gene is targeted by CREST. Ghosh suspects the CREST gene
might be turning on the production of chemicals called growth
factors, for the stimulatory effect they have on cell
development.
The CREST protein produced by that gene is made in several
regions of the brain immediately after birth. In adults, the
protein is produced in a region of the brain known as the
hippocampus, which plays an important role in learning and
memory. Because of this, Ghosh suspects that CREST may be
necessary for the storage of new memories and the ability to
learn. His laboratory is currently developing mice in which
CREST expression is normal throughout most of development, so
the brain develops normally, but then shuts off in the
hippocampus when the mice reach adulthood. In this way, the
researchers can test the specific role of CREST in learning and
memory in adults.
“Humans also have CREST, and the CREST gene sequence is highly
similar between mice and humans,” says Ghosh. “If it turns out
that CREST plays a role in learning and memory in the mouse,
then it is very likely it also plays a similar role in humans.”
The other researchers involved in the study are Hiroyuki Aizawa,
Shu-Ching Hu, Kathryn Bobb, Karthik Balakriashnan, Inga Gurevich
and Mitra Cowan. The study was supported by the National
Institutes of Health, the March of Dimes Birth Defects
Foundation, the Klingenstein Foundation, Merck and the Uehara
Memorial Foundation.
back to the top ~
back to Breaking News
~ back to
What's New
|